- Marketplace
- Merchants
- Restaurants
- UAE house
- Cloud accounting (GCC VAT)
- Me
- Cart
- My order
- Address book
- Favorite
- Link
- About us
- Download App
- 中文
- Merchant panel
CAS 120511-73-1 Anastrozole Anti Estrogen Steroids Raw Powder Arimidex
bargain
add to cartAnastrozole
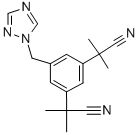
CAS:120511-73-1
MF:C17H19N5
MW:293.37
Synonyms:ICI-D-1033;ARIMIDEX;1-[3,5-DI-(1-METHYL-1-CYANO)-ETHYL]-BENZYL-1,2,4-TRIAZOLE;ANASTRAZOLE;ANASTROZOLE;tetramethyl-5-(1h-1,2,4-triazol-1ylmethyl) 1,3-benzenediacetionitrile;ZD-1033;1,3-benzenediacetonitrile,alpha,alpha,alpha' ,alpha' -tetramethyl-5-(1h-1,2,4-t
Properties
Melting point:81-82°C
storage temp. Store at RT
solubility DMSO: soluble40mg/mL
InChIKeyYBBLVLTVTVSKRW-UHFFFAOYSA-N
Safety Information
Symbol(GHS)


Description
Arimidex does not have the side effects of aminoglutethimide (Cytadren) and can achieve a high degree of estrogen blockade, much moreso than Cytadren. It is possible to reduce estrogen too much with Arimidex, and for this reason blood tests, or less preferably salivary tests, should be taken after the first week of use to determine if the dosing is correct.
As an aromatase inhibitor, Arimidex's mechanism of action - blocking conversion of aromatizable steroids to estrogen - is in contrast to the mechanism of action of anti-estrogens such as clomiphene (Clomid) or tamoxifen (Nolvadex), which block estrogen receptors in some tissues, and activate estrogen receptors in others. During a cycle, if using Arimidex, there is generally no need to use Clomid as well, but (as mentioned in the section on Clomid) there may still be benefits to doing so.
Arimidex at 0.5 mg/day is usually sufficient for moderate dosages of testosterone and in some cases may be too much.
Specifications
| Items | Standard | Results |
| Description | White crystalline powder | Complies |
| Identification | IR HPLC Reactive | Conform with standard IR Conform with standard HPLC Conform |
| Solubility | Freely soluble in methanol, acetone,ethanol and tetrahydrofuran. And very soluble in acetonitrile | Complies |
| Total Impurity | max.0.5% | 0.18% |
| Total Unspecified Impurity | max. 0.2% | 0.08% |
| Individual unspecified impurlty | max. 0.1% | 0.05% |
| Related compound B | max. 0.2% | 0.07% |
| Related compound C | max. 0.2% | 0.04% |
| Related compound D | max. 0.1% | 0.05% |
| Related compound E | max. 0.1% | 0.06% |
| Limit of cyclohexane | max. 0.08% | ND |
| Limit of ethylacetate | max. 0.1% | 0.07% |
| Melting point | 81.0~84.0ºC | 82.5~83.2ºC |
| Water | max. 0.3% | 0.21% |
| Residue on ignition | max. 0.1% | 0.07% |
| Heavy Metals | max.0.001% | Complies |
| Assay(HPLC) | 98.0%~102.0% | 99.8% |
| Storage | Cool and dry | |
| Conclusion | It complies to USP32 . | |
How ARIMIDEX works?
ARIMIDEX lowers the amount of oestrogen in the body. The female sex hormones oestrogen and progesterone stimulate many breast cancers to grow. Lowering the level of oestrogen can stop or slow the growth of breast cancer cells.
ARIMIDEX blocks a process called aromatisation which changes sex hormones called androgens into oestrogen. This happens mainly in the fatty tissues, muscle and the skin and needs a particular enzyme called aromatase.
Mechanism of Action:
The growth of many cancers of the breast is stimulated or maintained by estrogens.
In postmenopausal women, estrogens are mainly derived from the action of the aromatase enzyme, which converts adrenal androgens (primarily androstenedione and testosterone) to estrone and estradiol. The suppression of estrogen biosynthesis in peripheral tissues and in the cancer tissue itself can therefore be achieved by specifically inhibiting the aromatase enzyme.
ARIMIDEX is a selective non-steroidal aromatase inhibitor. It significantly lowers serum estradiol concentrations and has no detectable effect on formation of adrenal corticosteroids or aldosterone.
Adjuvant Treatment:
ARIMIDEX is indicated for adjuvant treatment of postmenopausal women with hormone receptor-positive early breast cancer.
First-Line Treatment:
ARIMIDEX is indicated for the first-line treatment of postmenopausal women with hormone receptor-positive or hormone receptor unknown locally advanced or metastatic breast cancer.
Second-Line Treatment:
ARIMIDEX is indicated for the treatment of advanced breast cancer in postmenopausal women with disease progression following tamoxifen therapy. Patients with ER-negative disease and patients who did not respond to previous tamoxifen therapy rarely responded to ARIMIDEX.
Dosage and direction:
Follow all instructions of your doctor about treatment and dosing schedule. Do not take more of this medication than it was administered to you. Take once daily.
Very discreet and exquisite packing methods to guarantee 100% custom pass rate(with more than 16 years experience).
All products are produced in Aseptic Laboratory under GMP conditions. Top quality is guaranteed.We have passed ISO9001 in 2008.
Skype&Whatsapp:86 13690838732
Email: ycsales12@ycphar.com
